DPMAS - A Comprehensive Therapy for Liver Failure
Author:Branding Date:2023-08-01
Background of liver failure
According to the statistical data, there are 2 million deaths caused by liver disease annually across the world, which is the eleventh-leading cause of death [1]. Liver failure is when your liver cannot function well to perform its vital duties such as removing toxins from the blood and producing bile to help you digest. Liver failure usually occurred due to liver damage which can be caused by many factors such as virus, alcohol, drugs, obesity etc. Liver failure can be a life-threatening emergency, and it may be acute or chronic. However, acute liver failure (ALF) is difficult to identify in its early stages, resulting in frequent delays in initiation of treatment.
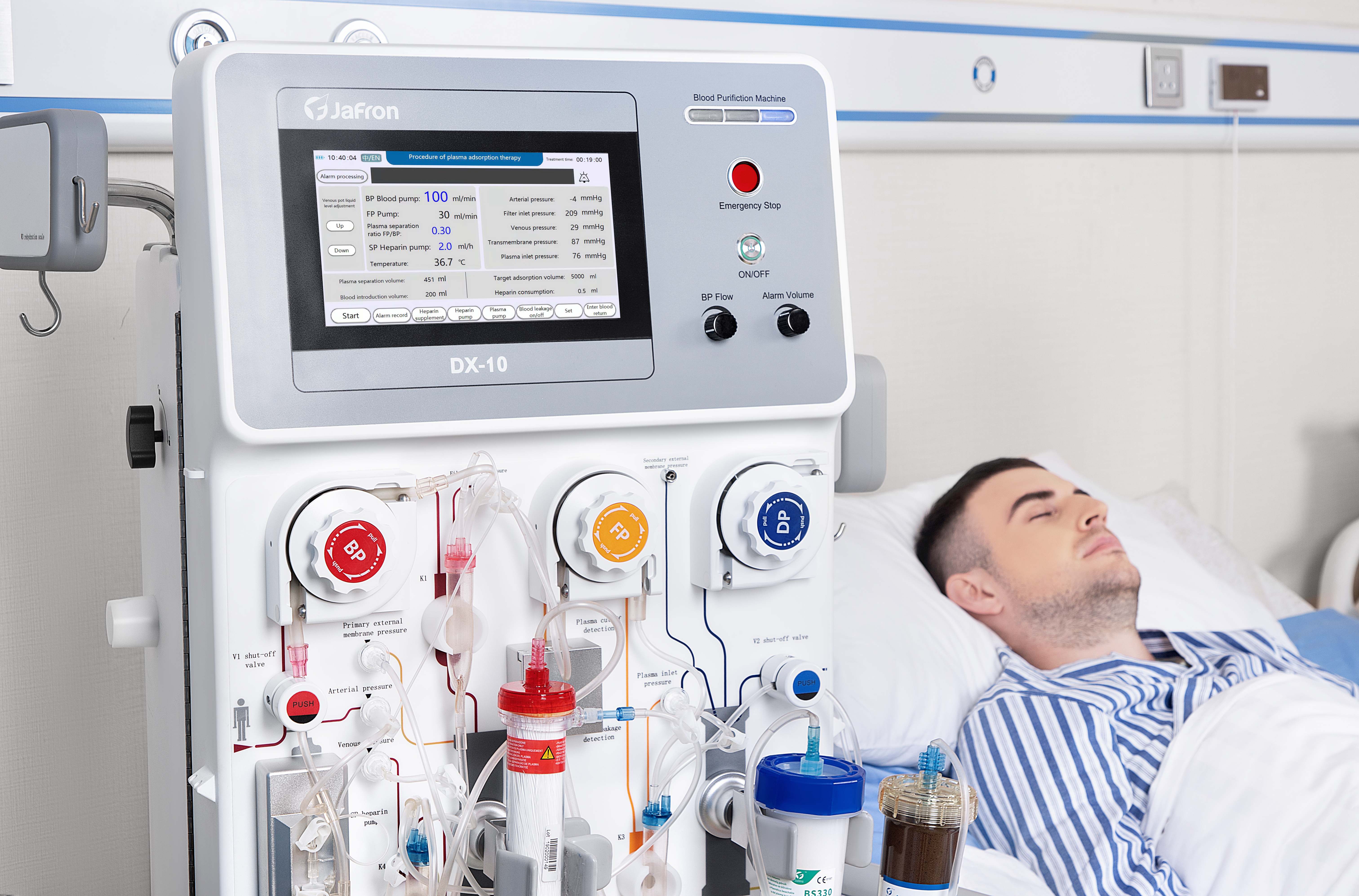
Treatment options for liver failure
Treatment options varies depending on the type of liver failure. For chronic liver failure (CLF), the treatment goal is mainly to slow and stop the progression of the disease and complications, such as lifestyle modification [2]. For acute liver failure (ALF), patient must be managed in the intensive care unit (ICU) and transferred to a center with a liver transplant facility due to its severity and complexity. The goal of ALF management is with aggressive supportive measures to avoid multi-organ failure until there is recovery or liver transplant [3]. During the last decades, research has found that the Artificial Liver Support System (ALSS) is effective, well tolerated and aimed at improving clinical, neurological, and biological parameters of patients with ALF. The most used ALSS including therapeutic plasma exchange (TPE), continuous hemodiafiltration, molecular absorbents recirculating system (MARS) and fractionated plasma separation and absorption (FPSA) [4]. However, these types of ALSS have shown some disadvantages and limitations. For instance, MARS and FPSA have failed to show improvement in survival in patients with ALF [5], and TPE has certain risk of infection, allergic reactions and ABO compatibility [6]. In that case, Double Plasma Molecular Adsorption System (DPMAS) is recommended as a new approach to relief the symptoms of patients with liver failure or its related complications.
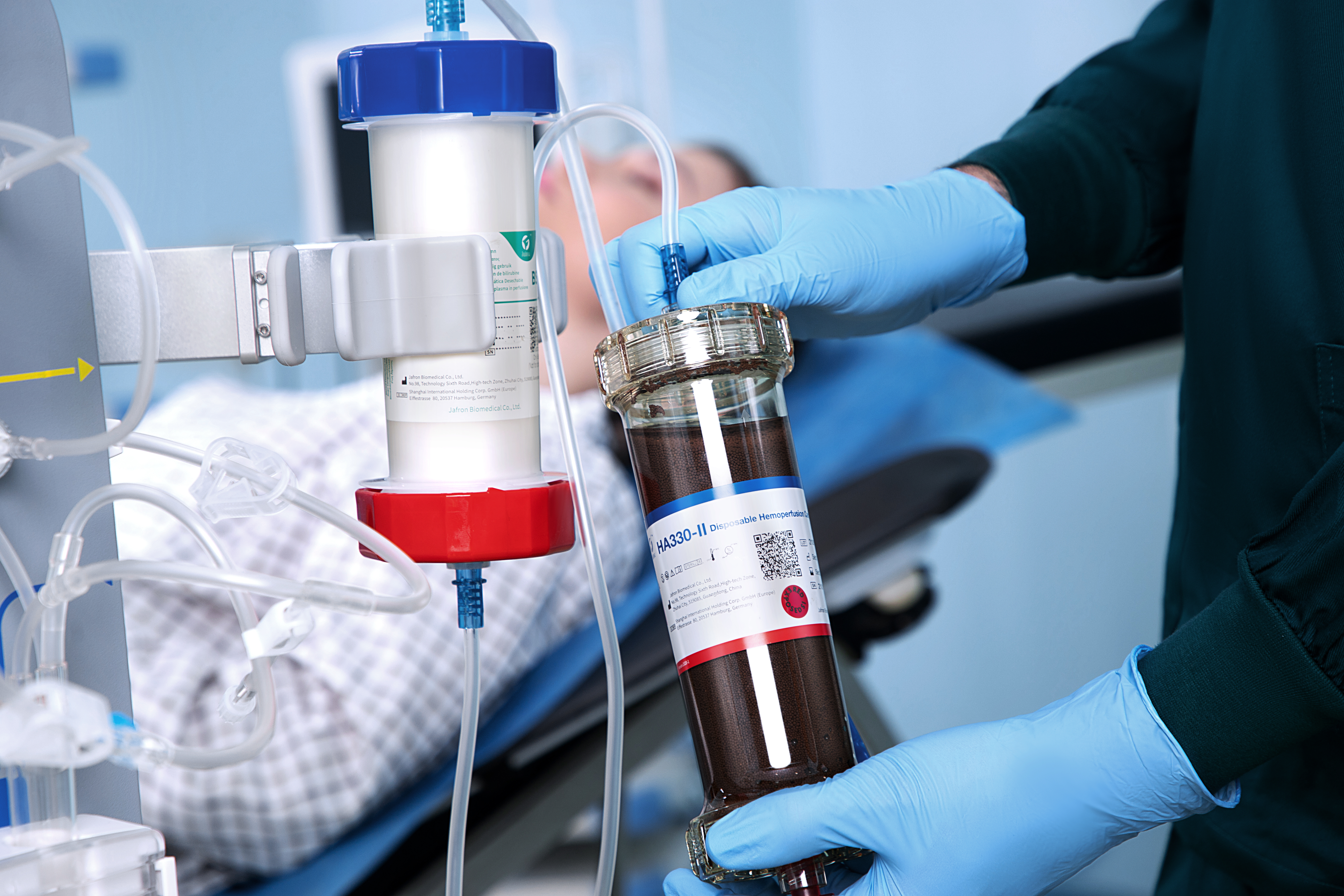
What is DPMAS?
DPMAS is a new therapy for liver disease. It adsorbs bilirubin, removes inflammatory mediators, eases inflammation and immune responses as well as considerably relieves the clinical symptoms and eventually improves the long-term prognosis of patients [7]. In brief, during the treatment, the plasma was separated, and then pass through the bilirubin adsorption column, BS330, which is filled up with anion-exchange resins, to adsorb bilirubin, and bile acid specifically. After that, the plasma will pass through HA330-II hemoadsorption cartridge to broad-spectrum adsorb toxins such as inflammatory mediators. The purified plasma will finally join up with the blood component and return to the patient.
Clinical application and benefits of DPMAS
DPMAS can be applied for patients with different conditions, including pre-, early, and mid-stage livers failures due to various reasons, pre and post liver transplant, severe hyperbilirubinemia, severe hyperbileacidemia or other disease with sever liver injury [8]. This mode of therapy does not require administration of blood products and can be performed even when plasma resources are limited. It also allows preservation of clotting factors or drugs. Most importantly, it is not associated with some adverse effects or allergies caused by exogenous plasma products [9].

In summary, DPMAS is a comprehensive and promising therapeutic approach for patients with liver failure. It has a strong ability to remove bilirubin, bile acids, inflammatory mediators, and medium to large molecular weight toxins, and can effectively delay the occurrence of MODS, while creating favorable conditions for liver function recovery, and rapidly improving jaundice symptoms in patients.
References
[1] Devarbhavi H, Asrani SK, et al. Global burden of liver disease: 2023 update. Journal of Hepatology. 2023;79(2):516-537. doi: 10.1016/j.jhep.2023.03.017
[2] Xu JH, Yu YY, Xu XY. Management of chronic liver diseases and cirrhosis: current status and future directions. Chin Med J (Engl). 2020 Nov 20;133(22):2647-2649. doi: 10.1097/CM9.0000000000001084
[3] Lopes D, Samant H. Hepatic Failure. [Updated 2023 Feb 12]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK538227/
[4] Tandon R, Froghi S. Artificial liver support systems. Journal of Gastroenterology and Hepatology, 2021;36: 1164– 1179. https://doi.org/10.1111/jgh.15255.
[5] Larsen FS. Artificial liver support in acute and acute-on-chronic liver failure. Current Option in Critical Care. 2019;25(2):187-191. doi: 10.1097/MCC.0000000000000584
[6] Afzali M, Oveisgharan S, Rajabkhah S, Abdi S. Complications of therapeutic plasma exchange in patients with neurological disorders. Curr J Neurol. 2020;19(1):8-12.
[7] Wu M, Zhang H, Huang Y, Wu W, Huang J, Yan D. Efficiency of Double Plasma Molecular Absorption System on the Acute Severe Cholestatic Hepatitis. Blood Purif. 2021;50(6):876-882. doi:10.1159/000513161
[8] Chen Y, Han T, Duan Z; Severe Liver Disease and Artificial Liver Group, Chinese Society of Hepatology, Chinese Medical Association. Clinical application of artificial liver and blood purification: expert consensus recommendations. Hepatol Int. 2023;17(1):4-17. doi:10.1007/s12072-022-10430-8
[9] Marcello M, Lorenzin A, De Cal M, et al. Bilirubin Removal by Plasmafiltration-Adsorption: Ex vivo Adsorption Kinetics Model and Single Case Report. Blood Purif. 2023;52(4):345-351. doi:10.1159/000528062
















